Hsc70 Antibody - #AF5187
| Product: | Hsc70 Antibody |
| Catalog: | AF5187 |
| Description: | Rabbit polyclonal antibody to Hsc70 |
| Application: | WB IHC IF/ICC |
| Reactivity: | Human, Mouse, Rat, Monkey |
| Prediction: | Pig, Zebrafish, Bovine, Horse, Sheep, Rabbit, Dog, Chicken |
| Mol.Wt.: | 70 kDa; 71kD(Calculated). |
| Uniprot: | P11142 |
| RRID: | AB_2837673 |
Related Downloads
Protocols
Product Info
*The optimal dilutions should be determined by the end user.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF5187, RRID:AB_2837673.
Fold/Unfold
2410008N15Rik; Constitutive heat shock protein 70; Epididymis luminal protein 33; Epididymis secretory sperm binding protein Li 72p; Heat shock 70 kDa protein 8; Heat shock 70kD protein 10; Heat shock 70kD protein 8; Heat shock 70kDa protein 8; Heat shock cognate 71 kDa protein; Heat shock cognate protein 54; Heat shock cognate protein 71 kDa; Heat shock protein 8; Heat shock protein A8; Heat-shock70-KD protein 10, formerly; HEL 33; HEL S 72p; HSC54; HSC71; Hsc73; HSP71; HSP73; HSP7C_HUMAN; HSPA10; HSPA8; LAP1; Lipopolysaccharide associated protein 1; LPS associated protein 1; LPS associated protein; MGC102007; MGC106514; MGC114311; MGC118485; MGC131511; MGC29929; N-myristoyltransferase inhibitor protein 71; NIP71;
Immunogens
- P11142 HSP7C_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MSKGPAVGIDLGTTYSCVGVFQHGKVEIIANDQGNRTTPSYVAFTDTERLIGDAAKNQVAMNPTNTVFDAKRLIGRRFDDAVVQSDMKHWPFMVVNDAGRPKVQVEYKGETKSFYPEEVSSMVLTKMKEIAEAYLGKTVTNAVVTVPAYFNDSQRQATKDAGTIAGLNVLRIINEPTAAAIAYGLDKKVGAERNVLIFDLGGGTFDVSILTIEDGIFEVKSTAGDTHLGGEDFDNRMVNHFIAEFKRKHKKDISENKRAVRRLRTACERAKRTLSSSTQASIEIDSLYEGIDFYTSITRARFEELNADLFRGTLDPVEKALRDAKLDKSQIHDIVLVGGSTRIPKIQKLLQDFFNGKELNKSINPDEAVAYGAAVQAAILSGDKSENVQDLLLLDVTPLSLGIETAGGVMTVLIKRNTTIPTKQTQTFTTYSDNQPGVLIQVYEGERAMTKDNNLLGKFELTGIPPAPRGVPQIEVTFDIDANGILNVSAVDKSTGKENKITITNDKGRLSKEDIERMVQEAEKYKAEDEKQRDKVSSKNSLESYAFNMKATVEDEKLQGKINDEDKQKILDKCNEIINWLDKNQTAEKEEFEHQQKELEKVCNPIITKLYQSAGGMPGGMPGGFPGGGAPPSGGASSGPTIEEVD
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
PTMs - P11142 As Substrate
| Site | PTM Type | Enzyme | Source |
|---|---|---|---|
| S2 | Acetylation | Uniprot | |
| S2 | Phosphorylation | Uniprot | |
| K3 | Acetylation | Uniprot | |
| K3 | Ubiquitination | Uniprot | |
| T13 | Phosphorylation | Uniprot | |
| Y15 | Phosphorylation | Uniprot | |
| S16 | Phosphorylation | Uniprot | |
| K25 | Ubiquitination | Uniprot | |
| T37 | Phosphorylation | Uniprot | |
| T38 | Phosphorylation | Uniprot | |
| S40 | Phosphorylation | Uniprot | |
| Y41 | Phosphorylation | Uniprot | |
| T45 | Phosphorylation | Uniprot | |
| T47 | Phosphorylation | Uniprot | |
| R49 | Methylation | Uniprot | |
| K56 | Acetylation | Uniprot | |
| K56 | Ubiquitination | Uniprot | |
| T64 | Phosphorylation | Uniprot | |
| T66 | Phosphorylation | Uniprot | |
| K71 | Acetylation | Uniprot | |
| K71 | Ubiquitination | Uniprot | |
| S85 | Phosphorylation | Uniprot | |
| K88 | Acetylation | Uniprot | |
| K88 | Methylation | Uniprot | |
| K88 | Ubiquitination | Uniprot | |
| K102 | Ubiquitination | Uniprot | |
| Y107 | Phosphorylation | Uniprot | |
| K108 | Acetylation | Uniprot | |
| K108 | Ubiquitination | Uniprot | |
| T111 | Phosphorylation | Uniprot | |
| K112 | Acetylation | Uniprot | |
| K112 | Ubiquitination | Uniprot | |
| S113 | Phosphorylation | Uniprot | |
| T125 | Phosphorylation | Uniprot | |
| K126 | Ubiquitination | Uniprot | |
| K128 | Acetylation | Uniprot | |
| K128 | Ubiquitination | Uniprot | |
| Y134 | Phosphorylation | Uniprot | |
| K137 | Acetylation | Uniprot | |
| K137 | Ubiquitination | Uniprot | |
| T138 | Phosphorylation | Uniprot | |
| T140 | Phosphorylation | Uniprot | |
| Y149 | Phosphorylation | Uniprot | |
| S153 | Phosphorylation | Uniprot | |
| T158 | Phosphorylation | Uniprot | |
| K159 | Acetylation | Uniprot | |
| K159 | Ubiquitination | Uniprot | |
| T163 | Phosphorylation | Uniprot | |
| T177 | Phosphorylation | Uniprot | |
| Y183 | Phosphorylation | Uniprot | |
| K187 | Acetylation | Uniprot | |
| K187 | Ubiquitination | Uniprot | |
| K188 | Ubiquitination | Uniprot | |
| K220 | Ubiquitination | Uniprot | |
| S221 | Phosphorylation | Uniprot | |
| T222 | Phosphorylation | Uniprot | |
| T226 | Phosphorylation | Uniprot | |
| K246 | Acetylation | Uniprot | |
| K246 | Methylation | Uniprot | |
| K246 | Ubiquitination | Uniprot | |
| K251 | Ubiquitination | Uniprot | |
| S254 | Phosphorylation | Uniprot | |
| K257 | Acetylation | Uniprot | |
| K257 | Sumoylation | Uniprot | |
| K257 | Ubiquitination | Uniprot | |
| T265 | Phosphorylation | Uniprot | |
| T273 | Phosphorylation | Uniprot | |
| S277 | Phosphorylation | Uniprot | |
| T298 | Phosphorylation | Uniprot | |
| T313 | Phosphorylation | Uniprot | |
| K319 | Acetylation | Uniprot | |
| K319 | Ubiquitination | Uniprot | |
| K325 | Ubiquitination | Uniprot | |
| K328 | Acetylation | Uniprot | |
| K328 | Ubiquitination | Uniprot | |
| S329 | Phosphorylation | Uniprot | |
| T341 | Phosphorylation | Uniprot | |
| K345 | Ubiquitination | Uniprot | |
| K348 | Acetylation | Uniprot | |
| K348 | Ubiquitination | Uniprot | |
| K357 | Acetylation | Uniprot | |
| K357 | Sumoylation | Uniprot | |
| K357 | Ubiquitination | Uniprot | |
| K361 | Acetylation | Uniprot | |
| K361 | Sumoylation | Uniprot | |
| K361 | Ubiquitination | Uniprot | |
| S362 | Phosphorylation | Uniprot | |
| Y371 | Phosphorylation | Uniprot | |
| S381 | Phosphorylation | Uniprot | |
| S400 | Phosphorylation | Uniprot | |
| T411 | Phosphorylation | Uniprot | |
| K415 | Ubiquitination | Uniprot | |
| T418 | Phosphorylation | Uniprot | |
| K423 | Ubiquitination | Uniprot | |
| T427 | Phosphorylation | Uniprot | |
| Y431 | Phosphorylation | Uniprot | |
| K451 | Acetylation | Uniprot | |
| K451 | Ubiquitination | Uniprot | |
| K458 | Ubiquitination | Uniprot | |
| T462 | Phosphorylation | Uniprot | |
| R469 | Methylation | Uniprot | |
| T477 | Phosphorylation | Uniprot | |
| K497 | Ubiquitination | Uniprot | |
| K500 | Ubiquitination | Uniprot | |
| T502 | Phosphorylation | Uniprot | |
| K507 | Acetylation | Uniprot | |
| K507 | Sumoylation | Uniprot | |
| K507 | Ubiquitination | Uniprot | |
| S511 | Phosphorylation | Uniprot | |
| K512 | Acetylation | Uniprot | |
| K512 | Sumoylation | Uniprot | |
| K512 | Ubiquitination | Uniprot | |
| K524 | Acetylation | Uniprot | |
| K524 | Ubiquitination | Uniprot | |
| Y525 | Phosphorylation | Uniprot | |
| K526 | Ubiquitination | Uniprot | |
| K531 | Ubiquitination | Uniprot | |
| K535 | Ubiquitination | Uniprot | |
| K539 | Acetylation | Uniprot | |
| K539 | Ubiquitination | Uniprot | |
| S541 | Phosphorylation | Uniprot | |
| S544 | Phosphorylation | Uniprot | |
| Y545 | Phosphorylation | Uniprot | |
| K550 | Ubiquitination | Uniprot | |
| K557 | Methylation | Uniprot | |
| K557 | Ubiquitination | Uniprot | |
| K561 | Acetylation | Uniprot | |
| K561 | Methylation | Uniprot | |
| K561 | Ubiquitination | Uniprot | |
| K567 | Methylation | Uniprot | |
| K569 | Methylation | Uniprot | |
| K573 | Ubiquitination | Uniprot | |
| C574 | S-Nitrosylation | Uniprot | |
| K583 | Acetylation | Uniprot | |
| K583 | Sumoylation | Uniprot | |
| K583 | Ubiquitination | Uniprot | |
| K589 | Acetylation | Uniprot | |
| K589 | Ubiquitination | Uniprot | |
| K597 | Acetylation | Uniprot | |
| K597 | Ubiquitination | Uniprot | |
| K601 | Acetylation | Uniprot | |
| K601 | Methylation | Uniprot | |
| K601 | Ubiquitination | Uniprot | |
| C603 | S-Nitrosylation | Uniprot | |
| T608 | Phosphorylation | Uniprot | |
| K609 | Ubiquitination | Uniprot | |
| S613 | Phosphorylation | Uniprot | |
| S637 | Phosphorylation | Uniprot | |
| S638 | Phosphorylation | Uniprot | |
| T641 | Phosphorylation | Uniprot |
Research Backgrounds
Molecular chaperone implicated in a wide variety of cellular processes, including protection of the proteome from stress, folding and transport of newly synthesized polypeptides, activation of proteolysis of misfolded proteins and the formation and dissociation of protein complexes. Plays a pivotal role in the protein quality control system, ensuring the correct folding of proteins, the re-folding of misfolded proteins and controlling the targeting of proteins for subsequent degradation. This is achieved through cycles of ATP binding, ATP hydrolysis and ADP release, mediated by co-chaperones. The co-chaperones have been shown to not only regulate different steps of the ATPase cycle of HSP70, but they also have an individual specificity such that one co-chaperone may promote folding of a substrate while another may promote degradation. The affinity of HSP70 for polypeptides is regulated by its nucleotide bound state. In the ATP-bound form, it has a low affinity for substrate proteins. However, upon hydrolysis of the ATP to ADP, it undergoes a conformational change that increases its affinity for substrate proteins. HSP70 goes through repeated cycles of ATP hydrolysis and nucleotide exchange, which permits cycles of substrate binding and release. The HSP70-associated co-chaperones are of three types: J-domain co-chaperones HSP40s (stimulate ATPase hydrolysis by HSP70), the nucleotide exchange factors (NEF) such as BAG1/2/3 (facilitate conversion of HSP70 from the ADP-bound to the ATP-bound state thereby promoting substrate release), and the TPR domain chaperones such as HOPX and STUB1. Acts as a repressor of transcriptional activation. Inhibits the transcriptional coactivator activity of CITED1 on Smad-mediated transcription. Component of the PRP19-CDC5L complex that forms an integral part of the spliceosome and is required for activating pre-mRNA splicing. May have a scaffolding role in the spliceosome assembly as it contacts all other components of the core complex. Binds bacterial lipopolysaccharide (LPS) and mediates LPS-induced inflammatory response, including TNF secretion by monocytes. Participates in the ER-associated degradation (ERAD) quality control pathway in conjunction with J domain-containing co-chaperones and the E3 ligase STUB1. Interacts with VGF-derived peptide TLQP-21.
Acetylated.
ISGylated.
Trimethylation at Lys-561 reduces fibrillar SNCA binding.
Cytoplasm. Melanosome. Nucleus>Nucleolus. Cell membrane.
Note: Localized in cytoplasmic mRNP granules containing untranslated mRNAs. Translocates rapidly from the cytoplasm to the nuclei, and especially to the nucleoli, upon heat shock.
Ubiquitous.
Identified in a IGF2BP1-dependent mRNP granule complex containing untranslated mRNAs. Interacts with PACRG. Interacts with HSPH1/HSP105. Interacts with IRAK1BP1 and BAG1. Interacts with DNAJC7. Interacts with DNAJB12 (via J domain). Interacts with DNAJB14 (via J domain). Interacts (via C-terminus) with the E3 ligase CHIP forming a 210 kDa complex of one CHIP and two HSPA8 molecules. Interacts with CITED1 (via N-terminus); the interaction suppresses the association of CITED1 to p300/CBP and Smad-mediated transcription transactivation. Component of the PRP19-CDC5L splicing complex composed of a core complex comprising a homotetramer of PRPF19, CDC5L, PLRG1 and BCAS2, and at least three less stably associated proteins CTNNBL1, CWC15 and HSPA8. Interacts with TRIM5. Part of a complex composed at least of ASCL2, EMSY, HCFC1, HSPA8, CCAR2, MATR3, MKI67, RBBP5, TUBB2A, WDR5 and ZNF335; this complex may have a histone H3-specific methyltransferase activity. Interacts with METTL21A. Following LPS binding, may form a complex with CXCR4, GDF5 and HSP90AA1. Interacts with PRKN. Interacts with FOXP3. Interacts with DNAJC9 (via J domain). Interacts with MLLT11. Interacts with RNF207. Interacts with DNAJC21. Interacts with DNAJB2. Interacts with TTC1 (via TPR repeats). Interacts with SGTA (via TPR repeats) (By similarity). Interacts with HSF1 (via transactivation domain). Interacts with HOPX, HSP40 and HSP90. Interacts with STUB1. Interacts with BAG2. Interacts with BAG3. Interacts with DNAJC12. Interacts with ZMYND10. Interacts with HSPC138. Interacts with BCL2L1, GIMAP5 and MCL1; the interaction with BCL2L1 or MCL1 is impaired in the absence of GIMAP5 (By similarity). Interacts with NLPR12. Interacts with TTC4.
(Microbial infection) Interacts with SV40 VP1.
The N-terminal nucleotide binding domain (NBD) (also known as the ATPase domain) is responsible for binding and hydrolyzing ATP. The C-terminal substrate-binding domain (SBD) (also known as peptide-binding domain) binds to the client/substrate proteins. The two domains are allosterically coupled so that, when ATP is bound to the NBD, the SBD binds relatively weakly to clients. When ADP is bound in the NBD, a conformational change enhances the affinity of the SBD for client proteins.
Belongs to the heat shock protein 70 family.
Research Fields
· Cellular Processes > Transport and catabolism > Endocytosis. (View pathway)
· Environmental Information Processing > Signal transduction > MAPK signaling pathway. (View pathway)
· Genetic Information Processing > Transcription > Spliceosome.
· Genetic Information Processing > Folding, sorting and degradation > Protein processing in endoplasmic reticulum. (View pathway)
· Human Diseases > Infectious diseases: Bacterial > Legionellosis.
· Human Diseases > Infectious diseases: Parasitic > Toxoplasmosis.
· Human Diseases > Infectious diseases: Viral > Measles.
· Human Diseases > Infectious diseases: Viral > Influenza A.
· Human Diseases > Infectious diseases: Viral > Epstein-Barr virus infection.
· Organismal Systems > Aging > Longevity regulating pathway - multiple species. (View pathway)
· Organismal Systems > Immune system > Antigen processing and presentation. (View pathway)
· Organismal Systems > Endocrine system > Estrogen signaling pathway. (View pathway)
References
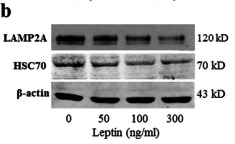
Application: WB Species: Rat Sample: rBMMSCs
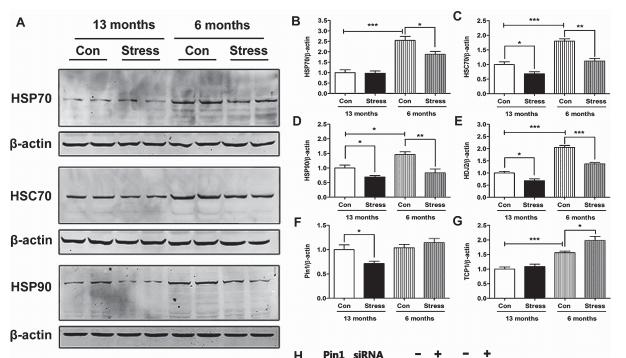
Application: WB Species: mouse Sample: mouse
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.