Fibrillin 1 Antibody - #AF0429
| Product: | Fibrillin 1 Antibody |
| Catalog: | AF0429 |
| Description: | Rabbit polyclonal antibody to Fibrillin 1 |
| Application: | WB IHC IF/ICC |
| Cited expt.: | WB, IHC |
| Reactivity: | Human, Mouse, Rat |
| Prediction: | Pig, Bovine, Horse, Sheep, Rabbit |
| Mol.Wt.: | 312kDa; 312kD(Calculated). |
| Uniprot: | P35555 |
| RRID: | AB_2834136 |
Related Downloads
Protocols
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF0429, RRID:AB_2834136.
Fold/Unfold
350 kDa glycoprotein component extracellular microfibril; ACMICD; FBN 1; FBN; FBN1; FBN1_HUMAN; Fibrillin 15; Fibrillin-1; Fibrillin1; Fibrillin15; GPHYSD2; Marfan syndrome; MASS; MFS 1; MFS1; OCTD; SGS; SSKS; Weill Marchesani syndrome; WMS; WMS2;
Immunogens
A synthesized peptide derived from human Fibrillin 1, corresponding to a region within C-terminal amino acids.
- P35555 FBN1_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MRRGRLLEIALGFTVLLASYTSHGADANLEAGNVKETRASRAKRRGGGGHDALKGPNVCGSRYNAYCCPGWKTLPGGNQCIVPICRHSCGDGFCSRPNMCTCPSGQIAPSCGSRSIQHCNIRCMNGGSCSDDHCLCQKGYIGTHCGQPVCESGCLNGGRCVAPNRCACTYGFTGPQCERDYRTGPCFTVISNQMCQGQLSGIVCTKTLCCATVGRAWGHPCEMCPAQPHPCRRGFIPNIRTGACQDVDECQAIPGLCQGGNCINTVGSFECKCPAGHKLNEVSQKCEDIDECSTIPGICEGGECTNTVSSYFCKCPPGFYTSPDGTRCIDVRPGYCYTALTNGRCSNQLPQSITKMQCCCDAGRCWSPGVTVAPEMCPIRATEDFNKLCSVPMVIPGRPEYPPPPLGPIPPVLPVPPGFPPGPQIPVPRPPVEYLYPSREPPRVLPVNVTDYCQLVRYLCQNGRCIPTPGSYRCECNKGFQLDLRGECIDVDECEKNPCAGGECINNQGSYTCQCRAGYQSTLTRTECRDIDECLQNGRICNNGRCINTDGSFHCVCNAGFHVTRDGKNCEDMDECSIRNMCLNGMCINEDGSFKCICKPGFQLASDGRYCKDINECETPGICMNGRCVNTDGSYRCECFPGLAVGLDGRVCVDTHMRSTCYGGYKRGQCIKPLFGAVTKSECCCASTEYAFGEPCQPCPAQNSAEYQALCSSGPGMTSAGSDINECALDPDICPNGICENLRGTYKCICNSGYEVDSTGKNCVDINECVLNSLLCDNGQCRNTPGSFVCTCPKGFIYKPDLKTCEDIDECESSPCINGVCKNSPGSFICECSSESTLDPTKTICIETIKGTCWQTVIDGRCEININGATLKSQCCSSLGAAWGSPCTLCQVDPICGKGYSRIKGTQCEDIDECEVFPGVCKNGLCVNTRGSFKCQCPSGMTLDATGRICLDIRLETCFLRYEDEECTLPIAGRHRMDACCCSVGAAWGTEECEECPMRNTPEYEELCPRGPGFATKEITNGKPFFKDINECKMIPSLCTHGKCRNTIGSFKCRCDSGFALDSEERNCTDIDECRISPDLCGRGQCVNTPGDFECKCDEGYESGFMMMKNCMDIDECQRDPLLCRGGVCHNTEGSYRCECPPGHQLSPNISACIDINECELSAHLCPNGRCVNLIGKYQCACNPGYHSTPDRLFCVDIDECSIMNGGCETFCTNSEGSYECSCQPGFALMPDQRSCTDIDECEDNPNICDGGQCTNIPGEYRCLCYDGFMASEDMKTCVDVNECDLNPNICLSGTCENTKGSFICHCDMGYSGKKGKTGCTDINECEIGAHNCGKHAVCTNTAGSFKCSCSPGWIGDGIKCTDLDECSNGTHMCSQHADCKNTMGSYRCLCKEGYTGDGFTCTDLDECSENLNLCGNGQCLNAPGGYRCECDMGFVPSADGKACEDIDECSLPNICVFGTCHNLPGLFRCECEIGYELDRSGGNCTDVNECLDPTTCISGNCVNTPGSYICDCPPDFELNPTRVGCVDTRSGNCYLDIRPRGDNGDTACSNEIGVGVSKASCCCSLGKAWGTPCEMCPAVNTSEYKILCPGGEGFRPNPITVILEDIDECQELPGLCQGGKCINTFGSFQCRCPTGYYLNEDTRVCDDVNECETPGICGPGTCYNTVGNYTCICPPDYMQVNGGNNCMDMRRSLCYRNYYADNQTCDGELLFNMTKKMCCCSYNIGRAWNKPCEQCPIPSTDEFATLCGSQRPGFVIDIYTGLPVDIDECREIPGVCENGVCINMVGSFRCECPVGFFYNDKLLVCEDIDECQNGPVCQRNAECINTAGSYRCDCKPGYRFTSTGQCNDRNECQEIPNICSHGQCIDTVGSFYCLCHTGFKTNDDQTMCLDINECERDACGNGTCRNTIGSFNCRCNHGFILSHNNDCIDVDECASGNGNLCRNGQCINTVGSFQCQCNEGYEVAPDGRTCVDINECLLEPRKCAPGTCQNLDGSYRCICPPGYSLQNEKCEDIDECVEEPEICALGTCSNTEGSFKCLCPEGFSLSSSGRRCQDLRMSYCYAKFEGGKCSSPKSRNHSKQECCCALKGEGWGDPCELCPTEPDEAFRQICPYGSGIIVGPDDSAVDMDECKEPDVCKHGQCINTDGSYRCECPFGYILAGNECVDTDECSVGNPCGNGTCKNVIGGFECTCEEGFEPGPMMTCEDINECAQNPLLCAFRCVNTYGSYECKCPVGYVLREDRRMCKDEDECEEGKHDCTEKQMECKNLIGTYMCICGPGYQRRPDGEGCVDENECQTKPGICENGRCLNTRGSYTCECNDGFTASPNQDECLDNREGYCFTEVLQNMCQIGSSNRNPVTKSECCCDGGRGWGPHCEICPFQGTVAFKKLCPHGRGFMTNGADIDECKVIHDVCRNGECVNDRGSYHCICKTGYTPDITGTSCVDLNECNQAPKPCNFICKNTEGSYQCSCPKGYILQEDGRSCKDLDECATKQHNCQFLCVNTIGGFTCKCPPGFTQHHTSCIDNNECTSDINLCGSKGICQNTPGSFTCECQRGFSLDQTGSSCEDVDECEGNHRCQHGCQNIIGGYRCSCPQGYLQHYQWNQCVDENECLSAHICGGASCHNTLGSYKCMCPAGFQYEQFSGGCQDINECGSAQAPCSYGCSNTEGGYLCGCPPGYFRIGQGHCVSGMGMGRGNPEPPVSGEMDDNSLSPEACYECKINGYPKRGRKRRSTNETDASNIEDQSETEANVSLASWDVEKTAIFAFNISHVSNKVRILELLPALTTLTNHNRYLIESGNEDGFFKINQKEGISYLHFTKKKPVAGTYSLQISSTPLYKKKELNQLEDKYDKDYLSGELGDNLKMKIQVLLH
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
Research Backgrounds
Structural component of the 10-12 nm diameter microfibrils of the extracellular matrix, which conveys both structural and regulatory properties to load-bearing connective tissues. Fibrillin-1-containing microfibrils provide long-term force bearing structural support. In tissues such as the lung, blood vessels and skin, microfibrils form the periphery of the elastic fiber, acting as a scaffold for the deposition of elastin. In addition, microfibrils can occur as elastin-independent networks in tissues such as the ciliary zonule, tendon, cornea and glomerulus where they provide tensile strength and have anchoring roles. Fibrillin-1 also plays a key role in tissue homeostasis through specific interactions with growth factors, such as the bone morphogenetic proteins (BMPs), growth and differentiation factors (GDFs) and latent transforming growth factor-beta-binding proteins (LTBPs), cell-surface integrins and other extracellular matrix protein and proteoglycan components. Regulates osteoblast maturation by controlling TGF-beta bioavailability and calibrating TGF-beta and BMP levels, respectively (By similarity). Negatively regulates osteoclastogenesis by binding and sequestering an osteoclast differentiation and activation factor TNFSF11. This leads to disruption of TNFSF11-induced Ca(2+) signaling and impairment of TNFSF11-mediated nuclear translocation and activation of transcription factor NFATC1 which regulates genes important for osteoclast differentiation and function. Mediates cell adhesion via its binding to cell surface receptors integrins ITGAV:ITGB3 and ITGA5:ITGB1. Binds heparin and this interaction has an important role in the assembly of microfibrils.
Hormone that targets the liver to increase plasma glucose levels. Secreted by white adipose tissue and circulates in the plasma. Acts in response to fasting and promotes blood glucose elevation by binding to the surface of hepatocytes. Promotes hepatocyte glucose release by activating the protein kinase A activity in the liver, resulting in rapid glucose release into the circulation.
Cleavage of N- and C-terminus by furin is required for incorporation into the extracellular matrix and assembly into microfibrils. The C-terminus, which corresponds to the Asprosin chain, was initially thought to constitute a propeptide. Fibrillin-1 and Asprosin chains are still linked together during the secretion from cells, but are subsequently separated by furin, an essential step for incorporation of Fibrillin-1 into the nascent microfibrils.
Forms intermolecular disulfide bonds either with other fibrillin-1 molecules or with other components of the microfibrils.
Secreted.
Note: Fibrillin-1 and Asprosin chains are still linked together during the secretion from cells, but are subsequently separated by furin (PubMed:24982166).
Secreted.
Note: Secreted into the plasma.
Secreted>Extracellular space>Extracellular matrix.
Belongs to the fibrillin family.
References
Application: WB Species: Human Sample:
Application: IHC Species: Rat Sample: kidneys
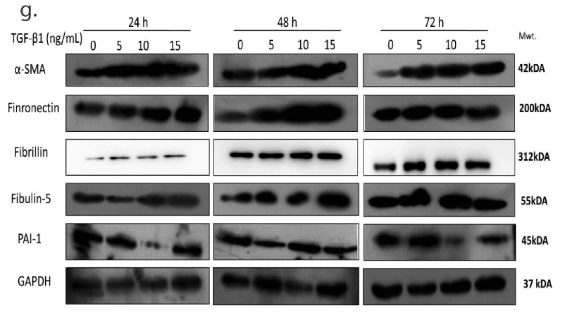
Application: WB Species: Human Sample: HTM Cells
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.