APP Antibody - #AF6084
| Product: | APP Antibody |
| Catalog: | AF6084 |
| Description: | Rabbit polyclonal antibody to APP |
| Application: | WB IHC IF/ICC |
| Cited expt.: | WB, IHC, IF/ICC |
| Reactivity: | Human, Mouse, Rat |
| Prediction: | Pig, Horse, Chicken, Xenopus |
| Mol.Wt.: | 117kDa; 87kD(Calculated). |
| Uniprot: | P05067 |
| RRID: | AB_2834853 |
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF6084, RRID:AB_2834853.
Fold/Unfold
A4_HUMAN; AAA; ABETA; ABPP; AD1; AICD-50; AICD-57; AICD-59; AID(50); AID(57); AID(59); Alzheimer disease amyloid protein; amyloid beta A4 protein; Amyloid intracellular domain 50; Amyloid intracellular domain 57; Amyloid intracellular domain 59; amyloid of aging and alzheimer disease; APP; APPI; beta-amyloid peptide; Beta-APP40; Beta-APP42; C31; Cerebral vascular amyloid peptide; CTFgamma; CVAP; Gamma-CTF(50); Gamma-CTF(57); Gamma-CTF(59); peptidase nexin-II; PN-II; PreA4; Protease nexin-II; S-APP-alpha; S-APP-beta;
Immunogens
A synthesized peptide derived from human APP, corresponding to a region within C-terminal amino acids.
Expressed in the brain and in cerebrospinal fluid (at protein level) (PubMed:2649245). Expressed in all fetal tissues examined with highest levels in brain, kidney, heart and spleen. Weak expression in liver. In adult brain, highest expression found in the frontal lobe of the cortex and in the anterior perisylvian cortex-opercular gyri. Moderate expression in the cerebellar cortex, the posterior perisylvian cortex-opercular gyri and the temporal associated cortex. Weak expression found in the striate, extra-striate and motor cortices. Expressed in cerebrospinal fluid, and plasma. Isoform APP695 is the predominant form in neuronal tissue, isoform APP751 and isoform APP770 are widely expressed in non-neuronal cells. Isoform APP751 is the most abundant form in T-lymphocytes. Appican is expressed in astrocytes.
- P05067 A4_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MLPGLALLLLAAWTARALEVPTDGNAGLLAEPQIAMFCGRLNMHMNVQNGKWDSDPSGTKTCIDTKEGILQYCQEVYPELQITNVVEANQPVTIQNWCKRGRKQCKTHPHFVIPYRCLVGEFVSDALLVPDKCKFLHQERMDVCETHLHWHTVAKETCSEKSTNLHDYGMLLPCGIDKFRGVEFVCCPLAEESDNVDSADAEEDDSDVWWGGADTDYADGSEDKVVEVAEEEEVAEVEEEEADDDEDDEDGDEVEEEAEEPYEEATERTTSIATTTTTTTESVEEVVREVCSEQAETGPCRAMISRWYFDVTEGKCAPFFYGGCGGNRNNFDTEEYCMAVCGSAMSQSLLKTTQEPLARDPVKLPTTAASTPDAVDKYLETPGDENEHAHFQKAKERLEAKHRERMSQVMREWEEAERQAKNLPKADKKAVIQHFQEKVESLEQEAANERQQLVETHMARVEAMLNDRRRLALENYITALQAVPPRPRHVFNMLKKYVRAEQKDRQHTLKHFEHVRMVDPKKAAQIRSQVMTHLRVIYERMNQSLSLLYNVPAVAEEIQDEVDELLQKEQNYSDDVLANMISEPRISYGNDALMPSLTETKTTVELLPVNGEFSLDDLQPWHSFGADSVPANTENEVEPVDARPAADRGLTTRPGSGLTNIKTEEISEVKMDAEFRHDSGYEVHHQKLVFFAEDVGSNKGAIIGLMVGGVVIATVIVITLVMLKKKQYTSIHHGVVEVDAAVTPEERHLSKMQQNGYENPTYKFFEQMQN
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
Research Backgrounds
Functions as a cell surface receptor and performs physiological functions on the surface of neurons relevant to neurite growth, neuronal adhesion and axonogenesis. Interaction between APP molecules on neighboring cells promotes synaptogenesis. Involved in cell mobility and transcription regulation through protein-protein interactions. Can promote transcription activation through binding to APBB1-KAT5 and inhibits Notch signaling through interaction with Numb. Couples to apoptosis-inducing pathways such as those mediated by G(O) and JIP. Inhibits G(o) alpha ATPase activity (By similarity). Acts as a kinesin I membrane receptor, mediating the axonal transport of beta-secretase and presenilin 1 (By similarity). By acting as a kinesin I membrane receptor, plays a role in axonal anterograde transport of cargo towards synapes in axons. Involved in copper homeostasis/oxidative stress through copper ion reduction. In vitro, copper-metallated APP induces neuronal death directly or is potentiated through Cu(2+)-mediated low-density lipoprotein oxidation. Can regulate neurite outgrowth through binding to components of the extracellular matrix such as heparin and collagen I and IV. The splice isoforms that contain the BPTI domain possess protease inhibitor activity. Induces a AGER-dependent pathway that involves activation of p38 MAPK, resulting in internalization of amyloid-beta peptide and leading to mitochondrial dysfunction in cultured cortical neurons. Provides Cu(2+) ions for GPC1 which are required for release of nitric oxide (NO) and subsequent degradation of the heparan sulfate chains on GPC1.
Amyloid-beta peptides are lipophilic metal chelators with metal-reducing activity. Bind transient metals such as copper, zinc and iron. In vitro, can reduce Cu(2+) and Fe(3+) to Cu(+) and Fe(2+), respectively. Amyloid-beta protein 42 is a more effective reductant than amyloid-beta protein 40. Amyloid-beta peptides bind to lipoproteins and apolipoproteins E and J in the CSF and to HDL particles in plasma, inhibiting metal-catalyzed oxidation of lipoproteins. APP42-beta may activate mononuclear phagocytes in the brain and elicit inflammatory responses. Promotes both tau aggregation and TPK II-mediated phosphorylation. Interaction with overexpressed HADH2 leads to oxidative stress and neurotoxicity. Also binds GPC1 in lipid rafts.
Appicans elicit adhesion of neural cells to the extracellular matrix and may regulate neurite outgrowth in the brain.
The gamma-CTF peptides as well as the caspase-cleaved peptides, including C31, are potent enhancers of neuronal apoptosis.
N-APP binds TNFRSF21 triggering caspase activation and degeneration of both neuronal cell bodies (via caspase-3) and axons (via caspase-6).
Proteolytically processed under normal cellular conditions. Cleavage either by alpha-secretase, beta-secretase or theta-secretase leads to generation and extracellular release of soluble APP peptides, S-APP-alpha and S-APP-beta, and the retention of corresponding membrane-anchored C-terminal fragments, C80, C83 and C99. Subsequent processing of C80 and C83 by gamma-secretase yields P3 peptides. This is the major secretory pathway and is non-amyloidogenic. Alternatively, presenilin/nicastrin-mediated gamma-secretase processing of C99 releases the amyloid-beta proteins, amyloid-beta protein 40 and amyloid-beta protein 42, major components of amyloid plaques, and the cytotoxic C-terminal fragments, gamma-CTF(50), gamma-CTF(57) and gamma-CTF(59). PSEN1 cleavage is more efficient with C83 than with C99 as substrate (in vitro). Many other minor amyloid-beta peptides, amyloid-beta 1-X peptides, are found in cerebral spinal fluid (CSF) including the amyloid-beta X-15 peptides, produced from the cleavage by alpha-secretase and all terminating at Gln-686.
Proteolytically cleaved by caspases during neuronal apoptosis. Cleavage at Asp-739 by either CASP6, CASP8 or CASP9 results in the production of the neurotoxic C31 peptide and the increased production of amyloid-beta peptides.
N-glycosylated. N- and O-glycosylated. O-glycosylation on Ser and Thr residues with core 1 or possibly core 8 glycans. Partial tyrosine glycosylation (Tyr-681) is found on some minor, short amyloid-beta peptides (amyloid-beta 1-15, 1-16, 1-17, 1-18, 1-19 and 1-20) but not found on amyloid-beta protein 38, amyloid-beta protein 40 nor on amyloid-beta protein 42. Modification on a tyrosine is unusual and is more prevelant in AD patients. Glycans had Neu5AcHex(Neu5Ac)HexNAc-O-Tyr, Neu5AcNeu5AcHex(Neu5Ac)HexNAc-O-Tyr and O-AcNeu5AcNeu5AcHex(Neu5Ac)HexNAc-O-Tyr structures, where O-Ac is O-acetylation of Neu5Ac. Neu5AcNeu5Ac is most likely Neu5Ac 2,8Neu5Ac linked. O-glycosylations in the vicinity of the cleavage sites may influence the proteolytic processing. Appicans are L-APP isoforms with O-linked chondroitin sulfate.
Phosphorylation in the C-terminal on tyrosine, threonine and serine residues is neuron-specific. Phosphorylation can affect APP processing, neuronal differentiation and interaction with other proteins. Phosphorylated on Thr-743 in neuronal cells by Cdc5 kinase and Mapk10, in dividing cells by Cdc2 kinase in a cell-cycle dependent manner with maximal levels at the G2/M phase and, in vitro, by GSK-3-beta. The Thr-743 phosphorylated form causes a conformational change which reduces binding of Fe65 family members. In dopaminergic (DA) neurons, phosphorylation on Thr-743 by LRKK2 promotes the production and the nuclear translocation of the APP intracellular domain (AICD) which induces DA neuron apoptosis. Phosphorylation on Tyr-757 is required for SHC binding. Phosphorylated in the extracellular domain by casein kinases on both soluble and membrane-bound APP. This phosphorylation is inhibited by heparin.
Extracellular binding and reduction of copper, results in a corresponding oxidation of Cys-144 and Cys-158, and the formation of a disulfide bond. In vitro, the APP-Cu(+) complex in the presence of hydrogen peroxide results in an increased production of amyloid-beta-containing peptides.
Trophic-factor deprivation triggers the cleavage of surface APP by beta-secretase to release sAPP-beta which is further cleaved to release an N-terminal fragment of APP (N-APP).
Amyloid-beta peptides are degraded by IDE.
Sulfated on tyrosine residues.
Cell membrane>Single-pass type I membrane protein. Membrane>Single-pass type I membrane protein. Perikaryon. Cell projection>Growth cone. Membrane>Clathrin-coated pit. Early endosome. Cytoplasmic vesicle.
Note: Cell surface protein that rapidly becomes internalized via clathrin-coated pits. Only a minor proportion is present at the cell membrane; most of the protein is present in intracellular vesicles (PubMed:20580937). During maturation, the immature APP (N-glycosylated in the endoplasmic reticulum) moves to the Golgi complex where complete maturation occurs (O-glycosylated and sulfated). After alpha-secretase cleavage, soluble APP is released into the extracellular space and the C-terminal is internalized to endosomes and lysosomes. Some APP accumulates in secretory transport vesicles leaving the late Golgi compartment and returns to the cell surface. APP sorts to the basolateral surface in epithelial cells. During neuronal differentiation, the Thr-743 phosphorylated form is located mainly in growth cones, moderately in neurites and sparingly in the cell body (PubMed:10341243). Casein kinase phosphorylation can occur either at the cell surface or within a post-Golgi compartment. Associates with GPC1 in perinuclear compartments. Colocalizes with SORL1 in a vesicular pattern in cytoplasm and perinuclear regions.
Secreted.
Cell surface.
Note: Associates with FPR2 at the cell surface and the complex is then rapidly internalized.
Nucleus. Cytoplasm.
Note: Located to both the cytoplasm and nuclei of neurons. It can be translocated to the nucleus through association with APBB1 (Fe65) (PubMed:11544248). In dopaminergic neurons, the phosphorylated Thr-743 form is localized to the nucleus (By similarity).
Expressed in the brain and in cerebrospinal fluid (at protein level). Expressed in all fetal tissues examined with highest levels in brain, kidney, heart and spleen. Weak expression in liver. In adult brain, highest expression found in the frontal lobe of the cortex and in the anterior perisylvian cortex-opercular gyri. Moderate expression in the cerebellar cortex, the posterior perisylvian cortex-opercular gyri and the temporal associated cortex. Weak expression found in the striate, extra-striate and motor cortices. Expressed in cerebrospinal fluid, and plasma. Isoform APP695 is the predominant form in neuronal tissue, isoform APP751 and isoform APP770 are widely expressed in non-neuronal cells. Isoform APP751 is the most abundant form in T-lymphocytes. Appican is expressed in astrocytes.
The transmembrane helix undergoes a conformation change and unravels partially when bound to PSEN1, facilitating cleavage by PSEN1.
The basolateral sorting signal (BaSS) is required for sorting of membrane proteins to the basolateral surface of epithelial cells.
The GFLD subdomain binds Cu(2+) ions; this promotes homodimerization.
The NPXY sequence motif found in many tyrosine-phosphorylated proteins is required for the specific binding of the PID domain. However, additional amino acids either N- or C-terminal to the NPXY motif are often required for complete interaction. The PID domain-containing proteins which bind APP require the YENPTY motif for full interaction. These interactions are independent of phosphorylation on the terminal tyrosine residue. The YENPXY site is also involved in clathrin-mediated endocytosis.
The C-terminal region can bind zinc ions; this favors dimerization and formation of higher oligomers.
The OX-2 motif shows some similarity to a region in the N-terminus of CD200/MOX2.
Belongs to the APP family.
Research Fields
· Human Diseases > Neurodegenerative diseases > Alzheimer's disease.
· Organismal Systems > Nervous system > Serotonergic synapse.
References
Application: IF/ICC Species: human Sample: HEK293 cells
Application: WB Species: human Sample: HEK293 cells
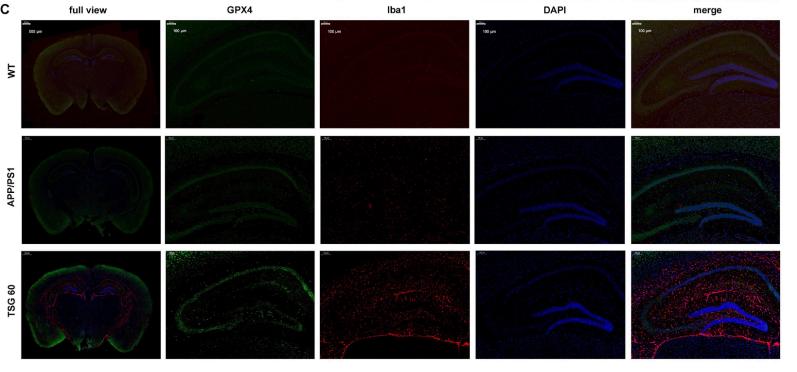
Application: IF/ICC Species: Mouse Sample: brain
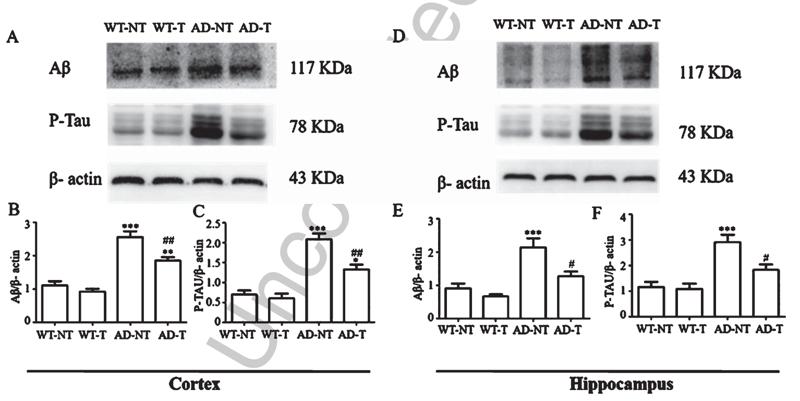
Application: WB Species: Mice Sample: Aβ25-35-induced HT22 cells
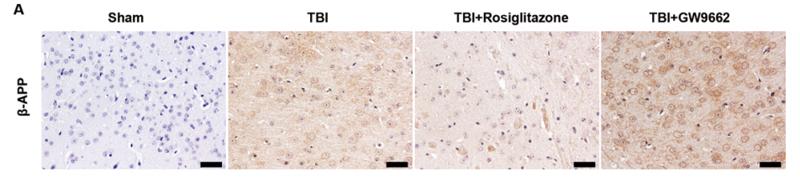
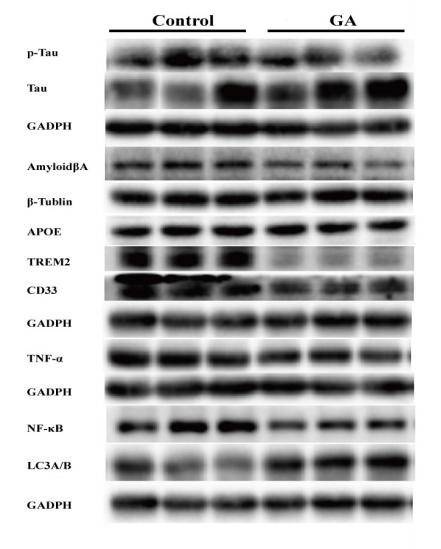
Application: WB Species: mouse Sample: brain
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.