VEGFR2 Antibody - #AF6281
| Product: | VEGFR2 Antibody |
| Catalog: | AF6281 |
| Description: | Rabbit polyclonal antibody to VEGFR2 |
| Application: | WB IHC IF/ICC |
| Cited expt.: | WB, IHC, IF/ICC |
| Reactivity: | Human, Mouse, Rat, Monkey |
| Prediction: | Pig, Bovine, Sheep, Rabbit, Dog |
| Mol.Wt.: | 180kDa; 152kD(Calculated). |
| Uniprot: | P35968 |
| RRID: | AB_2835132 |
Product Info
*The optimal dilutions should be determined by the end user. For optimal experimental results, antibody reuse is not recommended.
*Tips:
WB: For western blot detection of denatured protein samples. IHC: For immunohistochemical detection of paraffin sections (IHC-p) or frozen sections (IHC-f) of tissue samples. IF/ICC: For immunofluorescence detection of cell samples. ELISA(peptide): For ELISA detection of antigenic peptide.
Cite Format: Affinity Biosciences Cat# AF6281, RRID:AB_2835132.
Fold/Unfold
CD309; CD309 antigen; EC 2.7.10.1; Fetal liver kinase 1; FLK-1; FLK1; FLK1, mouse, homolog of; Kdr; Kinase insert domain receptor (a type III receptor tyrosine kinase); Kinase insert domain receptor; KRD1; Ly73; Protein tyrosine kinase receptor FLK1; Protein-tyrosine kinase receptor flk-1; soluble VEGFR2; Tyrosine kinase growth factor receptor; Vascular endothelial growth factor receptor 2; VEGFR 2; VEGFR; VEGFR-2; VEGFR2; VGFR2_HUMAN;
Immunogens
A synthesized peptide derived from human VEGFR2, corresponding to a region within the internal amino acids.
- P35968 VGFR2_HUMAN:
- Protein BLAST With
- NCBI/
- ExPASy/
- Uniprot
MQSKVLLAVALWLCVETRAASVGLPSVSLDLPRLSIQKDILTIKANTTLQITCRGQRDLDWLWPNNQSGSEQRVEVTECSDGLFCKTLTIPKVIGNDTGAYKCFYRETDLASVIYVYVQDYRSPFIASVSDQHGVVYITENKNKTVVIPCLGSISNLNVSLCARYPEKRFVPDGNRISWDSKKGFTIPSYMISYAGMVFCEAKINDESYQSIMYIVVVVGYRIYDVVLSPSHGIELSVGEKLVLNCTARTELNVGIDFNWEYPSSKHQHKKLVNRDLKTQSGSEMKKFLSTLTIDGVTRSDQGLYTCAASSGLMTKKNSTFVRVHEKPFVAFGSGMESLVEATVGERVRIPAKYLGYPPPEIKWYKNGIPLESNHTIKAGHVLTIMEVSERDTGNYTVILTNPISKEKQSHVVSLVVYVPPQIGEKSLISPVDSYQYGTTQTLTCTVYAIPPPHHIHWYWQLEEECANEPSQAVSVTNPYPCEEWRSVEDFQGGNKIEVNKNQFALIEGKNKTVSTLVIQAANVSALYKCEAVNKVGRGERVISFHVTRGPEITLQPDMQPTEQESVSLWCTADRSTFENLTWYKLGPQPLPIHVGELPTPVCKNLDTLWKLNATMFSNSTNDILIMELKNASLQDQGDYVCLAQDRKTKKRHCVVRQLTVLERVAPTITGNLENQTTSIGESIEVSCTASGNPPPQIMWFKDNETLVEDSGIVLKDGNRNLTIRRVRKEDEGLYTCQACSVLGCAKVEAFFIIEGAQEKTNLEIIILVGTAVIAMFFWLLLVIILRTVKRANGGELKTGYLSIVMDPDELPLDEHCERLPYDASKWEFPRDRLKLGKPLGRGAFGQVIEADAFGIDKTATCRTVAVKMLKEGATHSEHRALMSELKILIHIGHHLNVVNLLGACTKPGGPLMVIVEFCKFGNLSTYLRSKRNEFVPYKTKGARFRQGKDYVGAIPVDLKRRLDSITSSQSSASSGFVEEKSLSDVEEEEAPEDLYKDFLTLEHLICYSFQVAKGMEFLASRKCIHRDLAARNILLSEKNVVKICDFGLARDIYKDPDYVRKGDARLPLKWMAPETIFDRVYTIQSDVWSFGVLLWEIFSLGASPYPGVKIDEEFCRRLKEGTRMRAPDYTTPEMYQTMLDCWHGEPSQRPTFSELVEHLGNLLQANAQQDGKDYIVLPISETLSMEEDSGLSLPTSPVSCMEEEEVCDPKFHYDNTAGISQYLQNSKRKSRPVSVKTFEDIPLEEPEVKVIPDDNQTDSGMVLASEELKTLEDRTKLSPSFGGMVPSKSRESVASEGSNQTSGYQSGYHSDDTDTTVYSSEEAELLKLIEIGVQTGSTAQILQPDSGTTLSSPPV
Predictions
Score>80(red) has high confidence and is suggested to be used for WB detection. *The prediction model is mainly based on the alignment of immunogen sequences, the results are for reference only, not as the basis of quality assurance.
High(score>80) Medium(80>score>50) Low(score<50) No confidence
Research Backgrounds
Tyrosine-protein kinase that acts as a cell-surface receptor for VEGFA, VEGFC and VEGFD. Plays an essential role in the regulation of angiogenesis, vascular development, vascular permeability, and embryonic hematopoiesis. Promotes proliferation, survival, migration and differentiation of endothelial cells. Promotes reorganization of the actin cytoskeleton. Isoforms lacking a transmembrane domain, such as isoform 2 and isoform 3, may function as decoy receptors for VEGFA, VEGFC and/or VEGFD. Isoform 2 plays an important role as negative regulator of VEGFA- and VEGFC-mediated lymphangiogenesis by limiting the amount of free VEGFA and/or VEGFC and preventing their binding to FLT4. Modulates FLT1 and FLT4 signaling by forming heterodimers. Binding of vascular growth factors to isoform 1 leads to the activation of several signaling cascades. Activation of PLCG1 leads to the production of the cellular signaling molecules diacylglycerol and inositol 1,4,5-trisphosphate and the activation of protein kinase C. Mediates activation of MAPK1/ERK2, MAPK3/ERK1 and the MAP kinase signaling pathway, as well as of the AKT1 signaling pathway. Mediates phosphorylation of PIK3R1, the regulatory subunit of phosphatidylinositol 3-kinase, reorganization of the actin cytoskeleton and activation of PTK2/FAK1. Required for VEGFA-mediated induction of NOS2 and NOS3, leading to the production of the signaling molecule nitric oxide (NO) by endothelial cells. Phosphorylates PLCG1. Promotes phosphorylation of FYN, NCK1, NOS3, PIK3R1, PTK2/FAK1 and SRC.
N-glycosylated.
Ubiquitinated. Tyrosine phosphorylation of the receptor promotes its poly-ubiquitination, leading to its degradation via the proteasome or lysosomal proteases.
Autophosphorylated on tyrosine residues upon ligand binding. Autophosphorylation occurs in trans, i.e. one subunit of the dimeric receptor phosphorylates tyrosine residues on the other subunit. Phosphorylation at Tyr-951 is important for interaction with SH2D2A/TSAD and VEGFA-mediated reorganization of the actin cytoskeleton. Phosphorylation at Tyr-1175 is important for interaction with PLCG1 and SHB. Phosphorylation at Tyr-1214 is important for interaction with NCK1 and FYN. Dephosphorylated by PTPRB. Dephosphorylated by PTPRJ at Tyr-951, Tyr-996, Tyr-1054, Tyr-1059, Tyr-1175 and Tyr-1214.
The inhibitory disulfide bond between Cys-1024 and Cys-1045 may serve as a specific molecular switch for H(2)S-induced modification that regulates KDR/VEGFR2 function.
Cell junction. Endoplasmic reticulum.
Note: Localized with RAP1A at cell-cell junctions (By similarity). Colocalizes with ERN1 and XBP1 in the endoplasmic reticulum in endothelial cells in a vascular endothelial growth factor (VEGF)-dependent manner (PubMed:23529610).
Cell membrane>Single-pass type I membrane protein. Cytoplasm. Nucleus. Cytoplasmic vesicle. Early endosome.
Note: Detected on caveolae-enriched lipid rafts at the cell surface. Is recycled from the plasma membrane to endosomes and back again. Phosphorylation triggered by VEGFA binding promotes internalization and subsequent degradation. VEGFA binding triggers internalization and translocation to the nucleus.
Secreted.
Secreted.
Detected in cornea (at protein level). Widely expressed.
The second and third Ig-like C2-type (immunoglobulin-like) domains are sufficient for VEGFC binding.
Belongs to the protein kinase superfamily. Tyr protein kinase family. CSF-1/PDGF receptor subfamily.
Research Fields
· Cellular Processes > Cellular community - eukaryotes > Focal adhesion. (View pathway)
· Environmental Information Processing > Signal transduction > MAPK signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > Ras signaling pathway. (View pathway)
· Environmental Information Processing > Signal transduction > Rap1 signaling pathway. (View pathway)
· Environmental Information Processing > Signaling molecules and interaction > Cytokine-cytokine receptor interaction. (View pathway)
· Environmental Information Processing > Signal transduction > PI3K-Akt signaling pathway. (View pathway)
· Human Diseases > Drug resistance: Antineoplastic > EGFR tyrosine kinase inhibitor resistance.
· Human Diseases > Cancers: Overview > Proteoglycans in cancer.
References
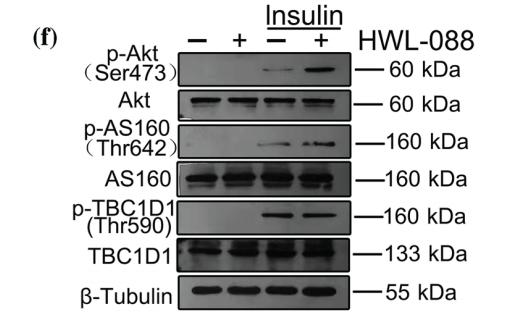
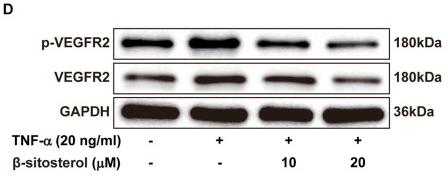
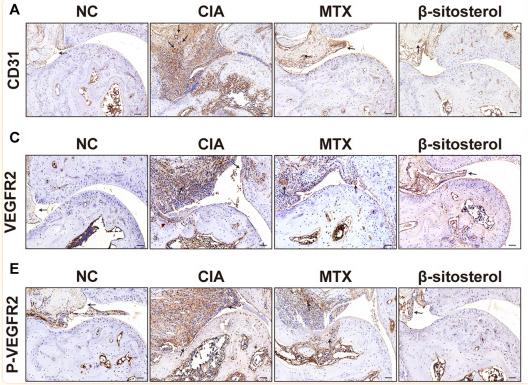
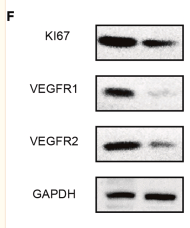
Application: WB Species: human Sample:
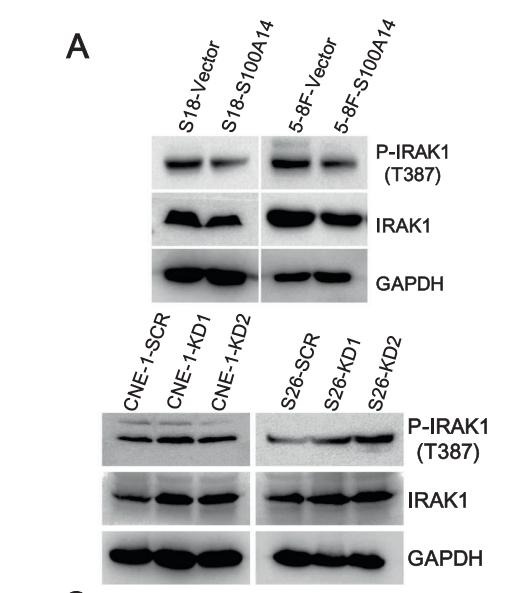
Application: WB Species: human Sample: A549 and H1299 cells
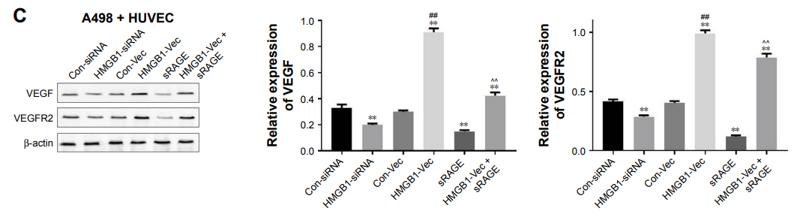
Application: WB Species: human Sample: PLC-PRF-5 cells
Application: IHC Species: human Sample: HCC
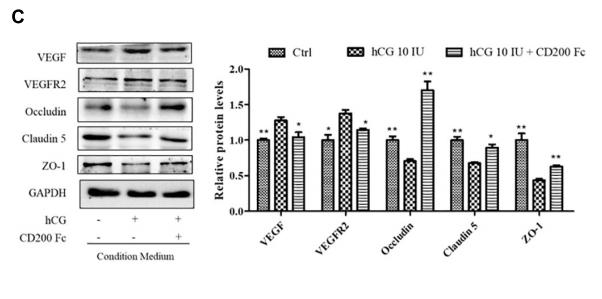
Application: IHC Species: Human Sample: ovarian tissue
Application: WB Species: human Sample: PLC-PRF-5 cells
Restrictive clause
Affinity Biosciences tests all products strictly. Citations are provided as a resource for additional applications that have not been validated by Affinity Biosciences. Please choose the appropriate format for each application and consult Materials and Methods sections for additional details about the use of any product in these publications.
For Research Use Only.
Not for use in diagnostic or therapeutic procedures. Not for resale. Not for distribution without written consent. Affinity Biosciences will not be held responsible for patent infringement or other violations that may occur with the use of our products. Affinity Biosciences, Affinity Biosciences Logo and all other trademarks are the property of Affinity Biosciences LTD.